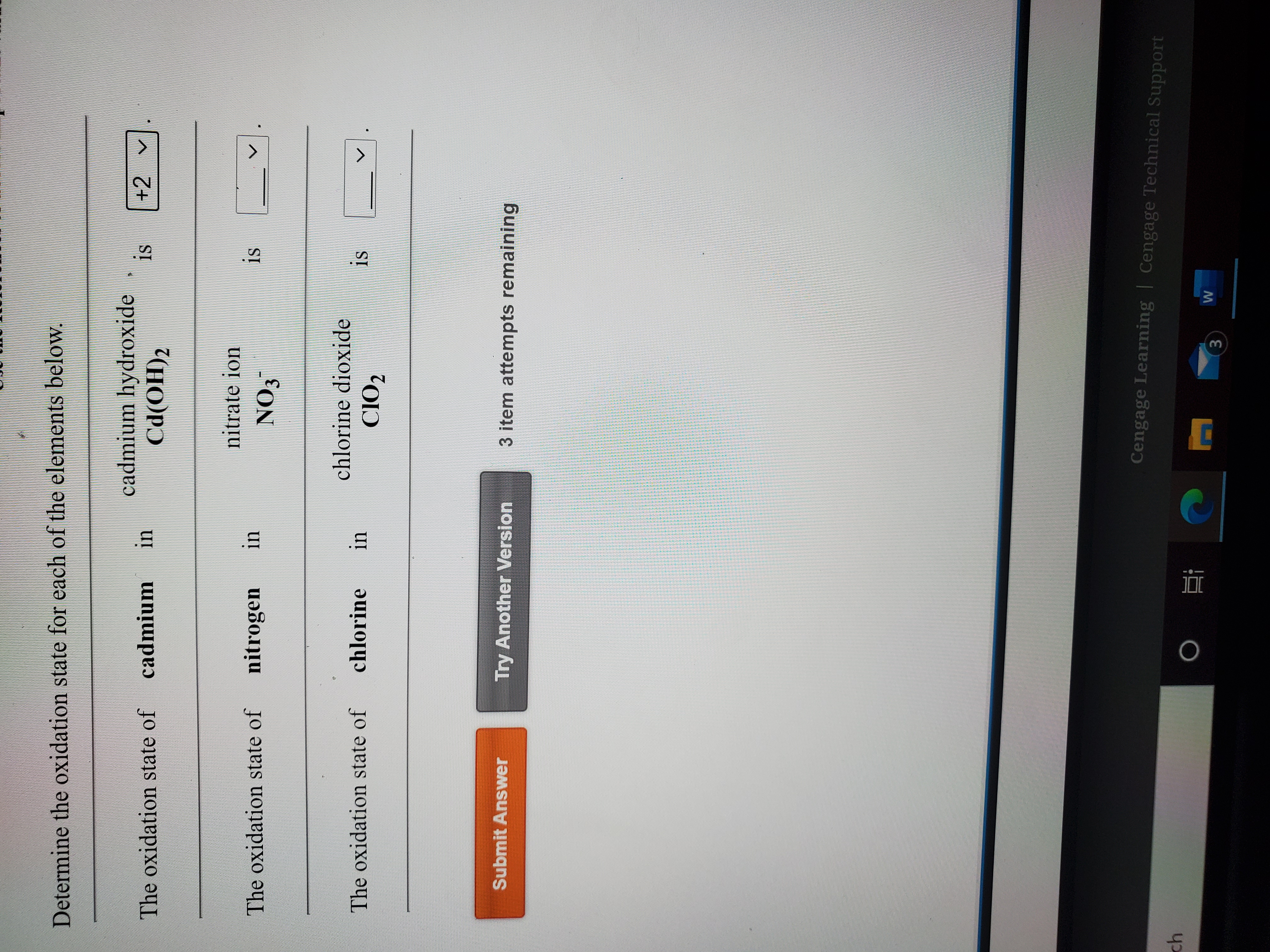
![What is the solubility of Cd(OH)2 in a buffer solution having pH=8 [Ksp of ( Cd(OH)2 is 2.5×10^-14]? - Brainly.in What is the solubility of Cd(OH)2 in a buffer solution having pH=8 [Ksp of ( Cd(OH)2 is 2.5×10^-14]? - Brainly.in](https://hi-static.z-dn.net/files/d7f/53875d97a1774f791777a981053518e7.jpg)
What is the solubility of Cd(OH)2 in a buffer solution having pH=8 [Ksp of ( Cd(OH)2 is 2.5×10^-14]? - Brainly.in

Solubility of `Cd(OH)_(2)` in pure water is `1.84xx10^(-5)\"mole\"//L` Calculate its solubility in - YouTube

SOLVED:Cd(OH)2 is an insoluble compound. It dissolves in excess NaOH in solution. Write a balanced ionic equation for this reaction. What type of reaction is this?

Synthesis of cadmium hydroxide nanostructure via composite-hydroxide-mediated approach - J Adnan, M Arfan, T Shahid, MZ Khan, R Masab, AH Ramish, S Ahtasham, AG Wattoo, M Hashim, A Zahoor, MF Nasir, 2019

Solubility of Cd(OH)(2) in pure water is 1.84xx10^(-5)"mole"//L Calculate its solubility in a buffer solution of ph=12.
The molar solubility of Cd(OH)2 is 1.84 × 10^–5 M in water. The expected solubility of Cd(OH)2 in a buffer solution of pH = 12 is : - Sarthaks eConnect | Largest Online Education Community